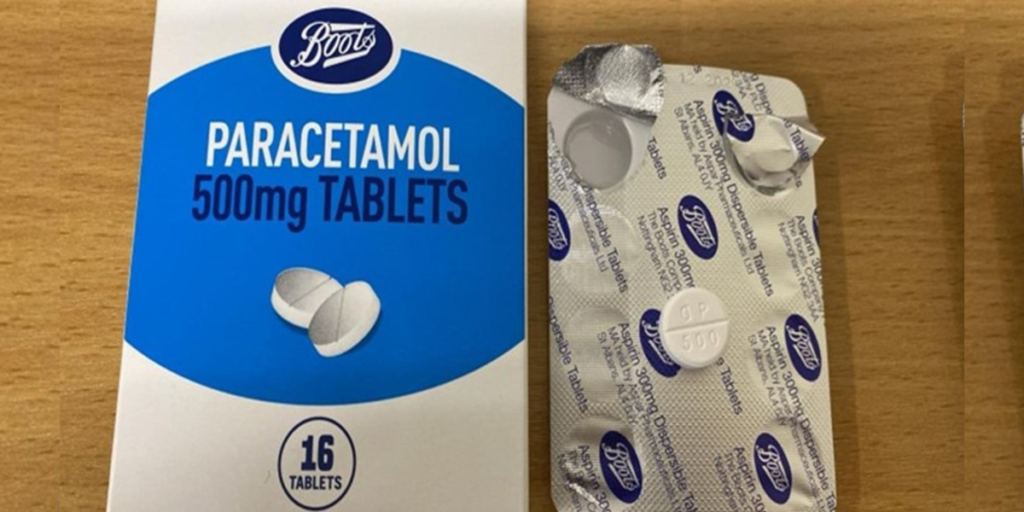
Boots, the UK’s beloved high-street pharmacy chain, has issued an urgent recall of its own-brand paracetamol tablets after a shocking labelling error misidentified the painkiller as aspirin, sending ripples of concern through customers and regulators alike. Announced on Tuesday, March 4, 2025, the Boots paracetamol recall targets batch number 241005 of its 500mg Tablets 16s, with the Medicines and Healthcare products Regulatory Agency (MHRA) warning users to “stop using immediately” and return the product for a full refund. As paracetamol recall news spreads, this blunder has thrust Boots into the spotlight, raising questions about safety, trust, and quality control in 2025’s pharmaceutical landscape.
The recall stems from a packaging snafu where the foil blister inside the carton reads “Aspirin 300mg Dispersible Tablets” instead of “Paracetamol 500mg Tablets,” despite confirmation that the tablets are, in fact, paracetamol. Affecting nearly 120,000 packs distributed since January 14, 2025, this paracetamol labelling error has sparked a frantic response from Boots and its supplier, Aspar Pharmaceuticals Limited, who are now scrambling to investigate how such a mix-up slipped through the cracks.
A Sudden Safety Scare
The Boots paracetamol recall hit social media on March 3, with the retailer posting a stark “do not take” alert on its Facebook page, urging customers to check their medicine cabinets. By Tuesday, the MHRA had escalated it to an official Class 2 Medicines Recall, emphasizing the risk of confusion. “It’s vitally important that you check the packaging,” said Dr. Stephanie Millican, MHRA Deputy Director of Benefit Risk Evaluation. “If the batch number is 241005, stop using the product and return it to a Boots store.”
This isn’t just a minor hiccup—it’s a potential hazard. While both paracetamol and aspirin are painkillers, their differences matter: aspirin can be dangerous for people with ulcers, asthma, or allergies, and dosing varies significantly. The paracetamol recall news has me reflecting on my own reliance on Boots as a go-to for quick fixes—headaches, colds, you name it. I’ve always trusted their shelves implicitly, so this feels like a personal jolt. How does a brand synonymous with British healthcare let this happen? It’s a question I suspect many are asking as they rummage through their drawers tonight.
Investigation Underway
Boots and Aspar Pharmaceuticals have confirmed the tablets are indeed paracetamol 500mg, not aspirin, but the labelling error alone is enough to warrant the recall. “The safety and well-being of our customers is very important to us,” Boots stated, offering refunds with or without receipts—a move to stem panic and reclaim trust. The paracetamol labelling error investigation is in full swing, with the supplier digging into how the wrong foil made it into production.
I can’t help but wonder about the human cost here. Someone, somewhere, missed this—and not just once, but across thousands of packs. As a journalist, I’ve seen recalls before—cheese with listeria, Biscoff with allergens—but this hits different. Paracetamol’s a household staple, not a niche treat. The Boots paracetamol recall could’ve been a disaster if someone allergic to aspirin had popped a pill, misled by the foil. It’s a sobering reminder that even giants can stumble, and it’s got me eyeing my own medicine stash with new skepticism.
Key Takeaways
- Boots Paracetamol Recall Alert: Batch 241005 of 500mg Tablets 16s is recalled due to a labelling error misidentifying it as aspirin.
- Safety First: The MHRA urges immediate return to Boots stores for a full refund, citing confusion risks despite correct contents.
- Massive Scale: Nearly 120,000 packs are affected, distributed across the UK since mid-January 2025.
- Ongoing Probe: Boots and Aspar Pharmaceuticals are investigating the paracetamol labelling error to prevent future mishaps.
Public Reaction and Trust at Stake
Posts on X are lighting up with reactions—some users joking about “aspirin roulette,” others fuming at Boots’ oversight. “Yet another error—these are happening too often,” one user lamented, echoing a sentiment of growing unease. The paracetamol recall news has also sparked practical pleas: check batch numbers (found near the barcode) and call Boots’ Customer Care at 0800 915 0004 with questions. For those who’ve taken the pills and felt off, the MHRA advises consulting a healthcare professional via its Yellow Card scheme.
This isn’t Boots’ first brush with scrutiny, but it’s a rare self-inflicted wound. I’ve walked those aisles since I was a kid—buying plasters, cough syrup, and yes, paracetamol—and there’s a comfort in that familiarity. Now, that trust feels wobbly. The paracetamol labelling error isn’t a contamination crisis, thank goodness, but it’s a crack in the facade of reliability. Will customers forgive and forget, or will this linger like a stubborn headache?
A Broader Context
This recall lands amid a busy 2025 news cycle—Trump’s fiery Congress speech, Al Green’s ejection, and market jitters over tariffs. Yet, the Boots paracetamol recall cuts through the noise because it’s personal. It’s not abstract policy or celebrity drama—it’s the pills in your cupboard. The MHRA’s swift action reflects a system on guard, but it also highlights how fragile that system can be. Past recalls—like nitrofurantoin capsules in 2024—show these glitches aren’t new, yet each one stings afresh.
For me, it’s a wake-up call about oversight in everyday essentials. I’m betting Boots bounces back—they’ve got deep roots and a loyal base—but they’ll need to tighten up fast. The paracetamol recall news might fade, but the lesson won’t: precision matters, especially when it’s your health on the line.
What’s Next for Boots?
The retailer hasn’t set an end date for returns, signaling an all-hands effort to mop up this mess. Customers are urged to act pronto—check that batch (241005, expiry 12/2029), ditch the pack, and grab a refund. Meanwhile, the paracetamol labelling error probe could reshape how Boots and Aspar handle quality control, with ripple effects across UK pharmacies.
As I write this, I’m struck by the irony: a painkiller causing a headache for a titan like Boots. The paracetamol recall news is a rare misstep for a chain that’s weathered wars, recessions, and retail shifts. They’ll survive this—I’d wager my last fiver on it—but it’s a humbling stumble. For now, the spotlight’s on, and Britain’s watching. Stay sharp, Boots—this one’s not over yet.